|
|
Original Article
| ||||||
| Shortened antibody screening interval has decreased the incidence of delayed hemolytic transfusion reactions | ||||||
| Nozomi Takano1, Hiroyasu Yasuda1, Masami Kikuchi1, Kinuyo Kawabata1, Chikako Takeuchi-Baba1, Satoshi Ono1, Takako Ono1, Keiji Minakawa1, Akiko Sugawara1, Saori Miura1, Kazuya Watanabe1, Maiko Abe1, Hiroe Suzuki1, Mao Watanabe1, Mutsumi Sasaki1, Kazuhiko Ikeda1, Kenneth E. Nollet1, Hitoshi Ohto1,2 | ||||||
|
1Department of Blood Transfusion and Transplantation Immunology, Fukushima Medical University Hospital, Fukushima, Japan 2Department of Advanced Cancer Immunotherapy, Fukushima Medical University, Fukushima, Japan | ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
[Print This Article] [Similar articles in PubMed] [Similar articles in Google Scholar] |
| How to cite this article |
| Takano N, Yasuda H, Kikuchi M, Kawabata K, Takeuchi-Baba C, Ono S, Ono T, Minakawa K, Sugawara A, Miura S, Watanabe K, Abe M, Suzuki H, Watanabe M, Sasaki M, Ikeda K, Nollet KE, Ohto H. Shortened antibody screening interval has decreased the incidence of delayed hemolytic transfusion reactions. Int J Blood Transfus Immunohematol 2018;8:100042Z02NT2018. |
|
ABSTRACT
| ||||||
|
Aims: To ascertain the effects of allowable sampling time prior to red cell transfusion when screening for alloantibodies, we compared antibody detection rates and frequencies of delayed hemolytic transfusion reactions (DHTRs) among patients drawn within two weeks versus patients drawn within one week of transfusion. Methods: Alloantibody screening for 32,601 patients from January 1997 through August 2006 was done within two weeks of transfusion, and for 44,896 patients from September 2006 through March 2017 was done within one week. Among transfusion recipients, 6,234 screened within two weeks and 8,066 screened within one week were evaluated for DHTR. Results: Alloantibodies were detected in 1.2% of cases screened within two weeks and in 1.3% of cases screened within one week, with no statistical difference. Anti-Fyb was more frequently detected (115.8 versus 39.9 per 100,000 patients) among those screened within one week (p <0.001); anti-Dia showed a similar trend, but without statistical significance (p =0.065), whereas anti-c was less frequently detected (p <0.05). The number of DHTRs observed was significantly lower among those screened within one week, with two cases (1 in 4,031) versus eight (1 in 779) among those screened within 2 weeks (p <0.05). Conclusion: By shortening allowable sampling period for red cell alloantibody screening from within two weeks to within one week of transfusion, the detection rate of clinically significant antibodies, except for anti-c, increased, and the frequency of DHTR recipients decreased. Keywords: Alloantibody, Clinically significant antibody, Delayed hemolytic transfusion reaction, Irregular antibody screening | ||||||
|
INTRODUCTION
| ||||||
|
Red blood cell (RBC) alloantibody screening is essential to prevent acute and delayed hemolytic transfusion reactions (DHTRs); any failure to detect alloantibodies places a recipient at risk. Early screening might precede alloantibody induction and late screening might miss alloantibody levels that have fallen below detection sensitivity [1]. Guidelines are intended to balance high detection sensitivity against the practical matter of being able to find units that are serologically and physiologically compatible [2]. In the UK, the British Committee for Standards in Haematology (BCSH) recommends that serological studies use patient samples collected no more than 3 days prior to the actual transfusion if a patient has been transfused or pregnant within the preceding 3 months, with allowance for samples up to 7 days prior for patients with no alloantibody history, no recent transfusion, and no recent pregnancy [3]. AABB and Dutch Blood Transfusion Guidelines also recommend antibody screening no more than 3 days before red cell transfusion for a patient who has been pregnant or transfused within the previous three months [4],[5], whereas the Canadian Society for Transfusion Medicine advises that specimens be collected within 96 hours of transfusion [3]. BCSH acknowledges, however, that scientific evidence is limited, and that their policy should be considered as a baseline only [3]. Thus, in this study, we aspired to analyze a large body of historical data in which differences might emerge between patients screened within 2 weeks versus 1 week of red cell transfusion. Any such differences might inform future pre-transfusion guidelines. | ||||||
|
MATERIALS AND METHODS
| ||||||
|
Patients
Pre-transfusion serological tests
During the 2W period, screening was performed by a combination of Sal-RT10’, Bromelain one-step (Bro), and PEG-potentiated indirect antiglobulin test (PEG-IAT). In Bromelain one-step technique, 2 drops of patient’s serum/plasma, 1 drop of Bromelain reagent (Wako Pure Chemical Industries, Osaka, Japan) were mixed and incubated for 15 minutes at 37OC, and agglutination was determined after centrifugation. Screening for the 1W cohort was done by Sal-IS and PEG-IAT methods. Resolve Panels A and B (Ortho Clinical Diagnostics) and/or PANOCELL-16 (Immucor Gamma, GA, USA) were used for antibody identification. We did not omit direct cross-matching, even when screening did not detect unexpected alloantibodies. Samples collected within 3 days of RBC transfusion for patients ever transfused or pregnant within 3 months were used for cross-matching by albumin-enhanced indirect antiglobulin testing (ALB-IAT) through June, 2004, and with low ionic strength solution-enhanced indirect antiglobulin testing (LISS-IAT) since July, 2004.
Clinical diagnosis of DHTR
Statistical analysis
| ||||||
RESULTS | ||||||
|
Alloantibody detection rate and antibody specificity
Comparison of frequency of DHTR
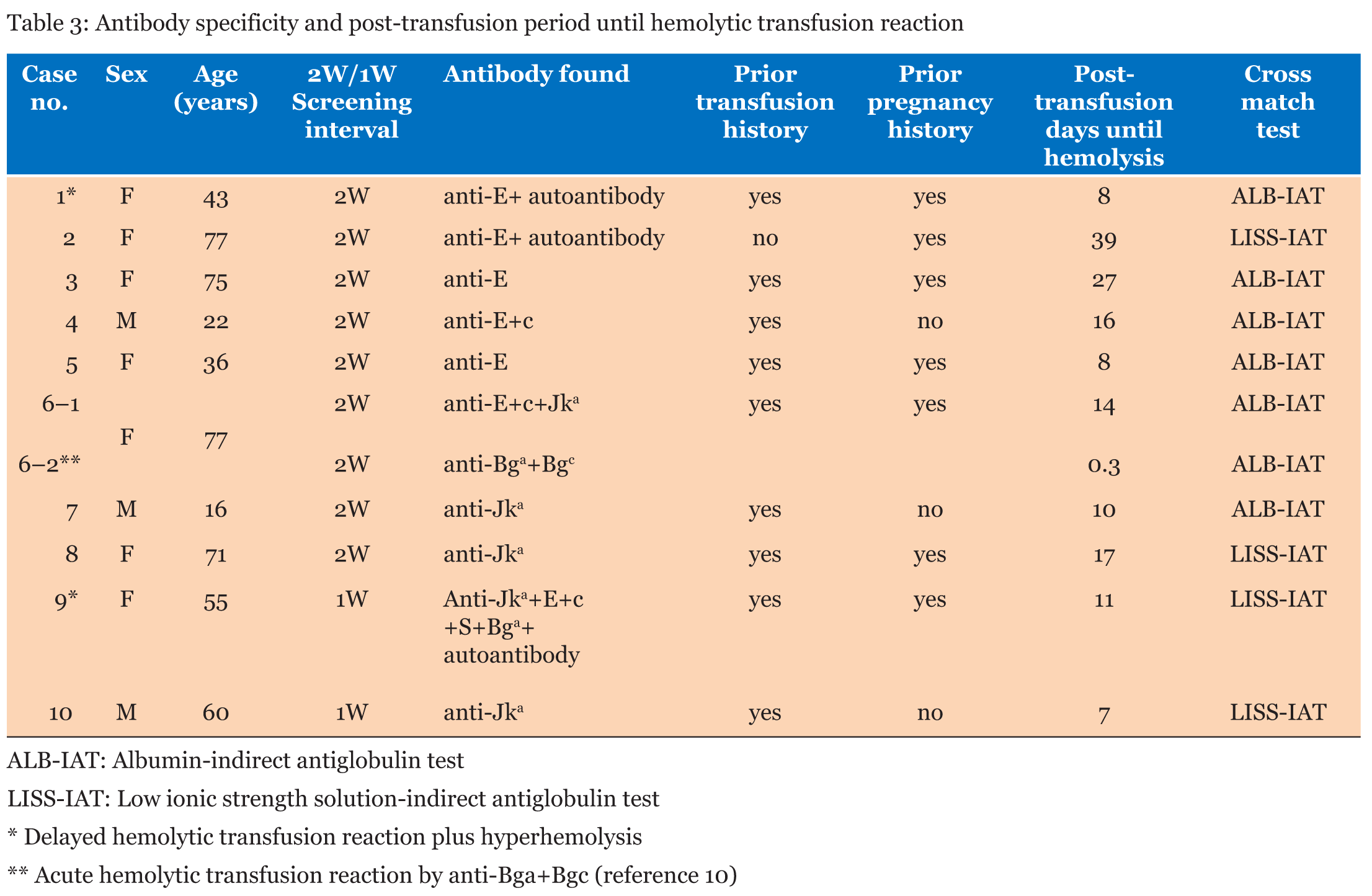
Table 3 shows the antibody specificity and the days from transfusion to hemolytic transfusion reaction for each case. The causative antibodies for hemolytic transfusion reactions were Rh in 5, Kidd in 3, Rh + Kidd in 2, and Bg in 1 cases. All of these patients had a history of transfusion or pregnancy. The time between RBC transfusion and DHTR (excluding acute hemolysis) ranged from 7 to 39 days. | ||||||
| ||||||
| ||||||
| ||||||
| ||||||
DISCUSSION | ||||||
|
There are insufficient data about the timing of alloantibodies formation and detection following red cell transfusion. Rarely, new antibodies are detected within the first 3 days, e.g. 0.4% (11 of 2,932 cases) or 2.3% of all immunized patients including secondary immune response [11]. Moreover, the majority (83%) of new antibodies are detected after more than 14 days, whereas only 17% are detected within 14 days after transfusion [11]. It is better established that most new antibodies manifest within the next 30 days, and very few antibodies develop after three months [3]. Japan’s National Health Insurance system has, since April 2006, agreed to cover alloantibody screening up to four times per month, versus twice per month prior to transfusion for patients who may have possibly transfusion. In order to investigate whether there are any beneficial effects of more frequent screening, with respect to alloantibody detection and frequency of DHTR, this retrospective, single-center, observational study was performed. In 2016, the Japan Society of Blood Transfusion and Cell Therapy advised that patients previously transfused or pregnant within the previous 3 months, should be cross-matched using samples no more than 3 days old at the time of transfusion [12]. Among clinically significant antibodies, we observed that the detection rate of anti-Fyb increased clearly and anti-Dia tended to increase within 1W versus 2W, probably because screening was done closer to the time of transfusion. It should be noted that PEG-IAT, reported to be more sensitive than other techniques, especially in detecting Kidd, Duffy, and Rh system antibodies [2], has been used as a standard screening test throughout both periods [6]. On the other hand, the detection frequency of anti-c decreased significantly from 101.2 within 2W to 51.2 per 100,000 within 1W (p <0.05). Anti-c is usually co-detected with anti-E, i.e. anti-E + c among Japanese, rather than emerging as a single antibody. This decreased anti-c frequency may be due to earlier detection of anti-E by a shortened sampling period and earlier selection of E-compatible RBCs for transfusion. Early anti-E detection within three days after transfusion has been observed in a previous study [11]. Among Japanese, the phenotype Rh Dee has a prevalence of 50%, consisting of DCCee at 43% and DCcee at 7% [13]. Therefore, when one bag of E-compatible RBCs is selected, 86% of the time it will lack c antigen; this serves as an incidental prophylaxis against the development of anti-c. Of note, the detection rates of cold antibodies, anti-Lea and anti-P1, significantly decreased (p <0.01, p <0.001). Because we changed screening from Sal-RT10’ plus Bro and PEG-IAT within 2W to Sal-IS and PEG-IAT within 1W, these cold-reactive antibodies were less often detected. When anti-Lea and anti-P1 results are negative with a potentiator-free indirect anti-globulin test, it has been suggested that these antigens can be ignored when selecting “compatible” RBCs [3], [4],[14] DHTRs observed in our study decreased significantly within 1W compared with 2W (p =0.045). In the past, DHTRs due to a primary immune response were often reported [15],[16] , but since then DHTRs have shifted predominantly to secondary immune responses [17], [18]. This shift might be generally attributed to technical changes, including the allowed sample collection period and test sensitivity. Also in this study, all patients who developed DHTRs had past transfusion and/or pregnancy history, which is compatible with a concept that many DHTRs can arise from a secondary immune response. Even if an antibody was previously detected after a past transfusion or pregnancy but falls below detection sensitivity in a current screening test, incompatible RBCs could evoke a DHTR. In our experience, the time until DHTR ranged from 7 to 39 days; therefore, with a 2 week screening interval, alloantibodies can be missed and incompatible RBCs might be administered. Nevertheless, while DHTRs are not completely preventable, by screening with the most sensitive methods such as PEG-IAT [2],[6] using samples drawn within one week of transfusion and cross-matching with samples drawn within three days of transfusion, DHTRs are decreasing. In the UK, where the vast majority of laboratories report through SHOT, a total of 18 cases of DHTR, including hyperhaemolysis, were documented in 2016, a number substantially lower than reported in previous years: 28 in 2015 and 2014, 32 in 2013, and 33 in 2012 [8]. Although approximately 80% are transfused within 72 hours of a new sample for screening and cross-matching being taken [19], BCSH speculates that this does not increase the number of additional DHTR cases being reported [3]. | ||||||
|
CONCLUSION
| ||||||
|
In conclusion with the decrease in the time allowed to collect alloantibody screening samples from within two weeks to within one week of transfusion, clinically significant antibody detection selectively increased while the frequency of DHTR decreased. | ||||||
|
REFERENCES
| ||||||
| ||||||
|
[HTML Abstract]
[PDF Full Text]
|
|
Author Contributions
Nozomi Takano – Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Hiroyasu Yasuda – Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Masami Kikuchi – Acquisition of data, Drafting the article, Final approval of the version to be published Kinuyo Kawabata – Acquisition of data, Analysis and interpretation of data, Drafting the article, Final approval of the version to be published Chikako Takeuchi-Baba – Acquisition of data, Drafting the article, Final approval of the version to be published Satoshi Ono – Acquisition of data, Drafting the article, Final approval of the version to be published Takako Ono – Acquisition of data, Drafting the article, Final approval of the version to be published Keiji Minakawa – Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Akiko Sugawara – Acquisition of data, Drafting the article, Final approval of the version to be published Saori Miura – Acquisition of data, Drafting the article, Final approval of the version to be published Kazuya Watanabe – Acquisition of data, Drafting the article, Final approval of the version to be published Maiko Abe – Acquisition of data, Drafting the article, Final approval of the version to be published Hiroe Suzuki – Acquisition of data, Drafting the article, Final approval of the version to be published Mao Watanabe – Acquisition of data, Drafting the article, Final approval of the version to be published Mutsumi Sasaki – Acquisition of data, Drafting the article, Final approval of the version to be published Kazuhiko Ikeda – Acquisition of data, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Kenneth E. Nollet – Acquisition of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published Hitoshi Ohto – Substantial contributions to conception and design, Analysis and interpretation of data, Drafting the article, Revising it critically for important intellectual content, Final approval of the version to be published |
|
Guarantor of Submission
The corresponding author is the guarantor of submission. |
|
Source of Support
None |
|
Consent Statement
Written informed consent was obtained from the patient for publication of this original article. |
|
Conflict of Interest
Author declares no conflict of interest. |
|
Copyright
© 2018 Nozomi Takano et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information. |
|
|