|
Case Report
Proactive strategies during a COVID-19 pandemic on regional center for blood transfusion in Oujda city and its impact on blood supply management
1 Centre Régional de Transfusion Sanguine, Avenue Errazi, Oujda 60000, Morocco
2 Centre National de Transfusion Sanguine et d’hématologie, 472, rue Mfadel Cherkaoui, q. Al Irfane, Rabat, Morocco
Address correspondence to:
Saliha Mharchi
Centre Régional de Transfusion Sanguine, Avenue Errazi, Oujda 60000,
Morocco
Message to Corresponding Author
Article ID: 100057Z02SM2021
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Mharchi S, Cherfi M, Karim A, Bouazza A, Sidqi Z, Benajiba M. Proactive strategies during a COVID-19 pandemic on regional center for blood transfusion in Oujda city and its impact on blood supply management. Int J Blood Transfus Immunohematol 2021;11:100057Z02SM2021.ABSTRACT
At the end of March 2020, the new coronavirus disease (COVID-19) caused by the SARS-CoV-2 virus was declared a global pandemic. By mid-May, the number of confirmed cases had risen to more than 5 million and more than 300,000 deaths were reported worldwide. Morocco confirmed its first imported case of COVID-19 on March 2, 2020. As of May 31, 2020, there were 7550 confirmed cases of infection with this virus. Like many countries, this epidemic prompted the Moroccan government, as of 20 March 2020, to apply travel restrictions and suspend travel within and between cities to prevent the spread of the disease. These distancing measures have resulted in effective control of the virus. However, they also have a profound effect on the health system, relatively speaking, on blood donation and supply and the management of transfusion at the national level. In this context, on March 20, 2020, WHO provided guidance on the management of the blood supply. In addition, the National Centre for Blood Transfusion and Haematology (NCBTH) has accompanied the Regional Centers for Blood Transfusion (RCBT) of the kingdom through the implementation of a new strategy to deal with a possible shortage of blood supply. In response to such a situation, the Oujda Regional Center for Blood Transfusion (ORCBT) anticipated the development of a proactive regional strategy in collaboration with the Blood Donor Association of the Eastern Region (BDAER), and set up accompanying measures during the pandemic. The objective of this study is to describe the strategies undertaken during the COVID-19 pandemic and their impact on all transfusion activities from blood collection, delivery, and transfusion of Labile Blood Products (LBPs). To achieve our objective, we performed a comparative prospective analytical study from January 1 to May 31 of 2019 and 2020 years at the ORCBT, located in the prefecture of Oujda Angad. The results of this study showed a positive impact, to some extent, of the strategies implemented, first on blood collection and then on the center′s safety stock, which remained at the same level despite the containment circumstances. Thus, the ORCBT carried out its mission by responding to the majority of requests for LBPs that were addressed to it with a satisfaction rate of over 90% for all health facilities in the eastern region during the study period.
Keywords: Blood demand, Blood donation, Blood transfusion, COVID-19, Eastern region, Label Blood Product
Introduction
Coronavirus 2 (COVID-19), a highly contagious respiratory disease caused by the SARS-CoV-2 virus, was born in Wuhan, China, in late 2019 [1]. On January 30, 2020, WHO declared this infection a public health emergency of international concern (PHEIC). As of February 11, 2020, the virus had affected 42,708 people in China and had spread to 25 countries, and since June 25, 2020, 216 countries have been affected [2]. In Africa, the first case was registered in Egypt on February 14, 2020 [3] and since June 24, 2020, more than 236,909 cases of COVID-19 have been confirmed with 5257 deaths [4]. As soon as the number of cases outside China increased 13-fold and the number of countries with cases tripled, WHO declared the new coronavirus (COVID-19) a global pandemic on March 11, 2020 [5]. Up to May 22, 2020, more than 100,000 cases of COVID-19 have been confirmed in Africa. Early statistics have shown that most of the first recorded cases were imported from European countries [6],[7].
In the early stages of a new epidemic, epidemiological information on the infection is often incomplete, including knowledge about the transmission of the disease. Nevertheless, the route of infection of COVID-19 is mainly person-to-person [8]. Although the virus has a preferential tropism for bronchial epithelial cells, there is evidence that viral RNA can be detected in the plasma of patients with low COVID-19 viral load [9],[10]. One study described cases of patients with very severe anemia who received apheresis platelets transfusion from an individual who was subsequently diagnosed with SARS-CoV-2 and the transfused patients tested negative for SARS-CoV-2 [11]. For this reason, blood establishments such as Transfusion Swiss Red Cross (SRC), the American Association of Blood Bank (AABB), and the Food and Drug Administration (FDA) do not currently require additional measures for the collection and testing of donations [12]. However, there is still a theoretical risk of transmission of COVID-19 by blood transfusion. As more and more asymptomatic infections are found among COVID-19 cases, blood safety and considerations of preventive measures that are likely to affect blood donation, particularly in endemic areas, need to be taken into consideration.
Highly detailed plans, such as those published by the AABB and Canada, had never been implemented before in response to a pandemic [13],[14]. However, even the best-prepared blood banks had to work diligently to make changes to operational plans when the COVID-19 pandemic was declared. Laboratories have even begun to develop techniques and new procedures for bacteriological testing to extend the expiration of platelets by 5 to 7 days [15]. With respect to all of the above, and to mitigate a potential shortage of blood supply, the WHO has provided guidance on the management of the blood supply [16],[17].
In Morocco, the first case of COVID-19 coronavirus was recorded on March 2, 2020 [18]. At the end of May, there were 7433 cases [19]. This growing evidence forced authorities and governments to declare a state of public health emergency on March 20, 2020, and implement all early protective measures to limit contamination and reduce the formation of new sources of contagion [18]: Individual confinement, suspension of international flights, suspension of classes at schools and universities, ban on meetings of more than 50 people, closure of public places (cafes, restaurants, cinemas, sports halls, and mosques). These distancing measures have made it possible, more or less, to control the spread of the virus. However, they also have a profound effect on the health system, including blood donor recruitment, blood supply and transfusion management at the national and regional levels. In addition to the limited lifespan of blood products, which complicates the management of LBPs supply in critical situations, strictly applied mobility restrictions put enormous pressure on the Regional Centers for Blood Transfusion (RCBT) to satisfy all the needs of healthcare institutions. In response to such a situation, the RCBT of Oujda anticipated the establishment of a proactive regional strategy based on the recommendations defined and implemented by the National Centre for Blood Transfusions and Haematology (NCBTH) at the national level [20].
This work describes the proactive measures and recommendations of the NCBTH implemented at the Oujda city RCBT in response to the COVID-19 pandemic and therefore analyze these impacts on all transfusion activities from blood collection, delivery, and transfusion of blood products.
MATERIALS AND METHODS
A 5-month cross-sectional study for descriptive purposes carried out at the RCBT in Oujda between January 1 and May 31 of both years 2019 and 2020. The data were progressively exploited and analyzed using the RCBT’s statistical database from the national computer system (e-Progesa) to achieve our research objectives. Ethical considerations were respected throughout the work, with anonymity and the approval of the Chief Doctor of the ORCBT and the Director of the NCBTH.
RESULTS
With the advent of this new coronavirus at the end of December 2019 and its impact on global health, the ORCBT anticipated the development of a proactive strategy to avoid any stockouts or shortages of LBPs during this pandemic period. Moreover, since March 20, 2020, the first day of the restrictive measures imposed by the Moroccan authorities, public and private institutions have called for limiting their activities or even suspending them depending on the vitality of the sector [18]. According to the NCBTH, the ORCBT has always been considered one of the basic reserves for other regional centers in the kingdom in case of LBP shortages. Thus, whatever the situation, the activity of the center of Oujda should not be interrupted.
The ORCBT’s prevention strategy to ensure self-sufficiency in LBPs
In the perspective of ensuring a permanent service and since February, the management committee of the Oujda center in collaboration with the BDAER has proceeded to organize regular meetings to study all possible eventualities and has made a series of recommendations to enhance the safety stock and ensure self-sufficiency in LBPs.
Proactive measures:
- Strengthening mobile blood drives: increasing the number of mobile blood drives and focus on sites with more than 100 donors per clinic.
- Raising the awareness of hospitals on the possible crisis forecast and encouraging them to rationalize their demand for LBPs and limit the number of LBPs to two LBPs per demand.
- Recruit donors through social media.
NCBTH pandemic strategy for COVID-19
The NCBTH is a national scientific reference. It is responsible for the implementation of the Ministry of Health’s policy on blood transfusion. Since the outbreak of the SARS-CoV-2 epidemic on December 31, 2019 in China, and in response to the guidelines of international organizations (WHO, ECDC, HCSP, and AABB), a set of recommendations have been addressed to the Kingdom’s RCBT on a regular basis in the field of blood transfusion and hematology. Thus, the National Center has guaranteed the continuous updating of the measures taken with regard to the risk of this new coronavirus in order to adapt its activities for a good management of the impact of the COVID-19 pandemic on blood donation both at the national level and at the level of the Regional Blood Transfusion Centers [20]. In addition to the measures implemented by the RCBT of Oujda, the directives issued by the NCBTH gave a legal foundation for its actions and encouraged the collaboration of the regional health department in Oujda in its actions. In addition, the NCBTH has regularly provided the RCBT of Oujda with disinfectant and antiseptic products and individual protection equipment to ensure the protection of donors and the professionals of the center. The recommendations of the NCBTH focused on the following areas:
Donor hemovigilance
- Strengthening pre-donation interview.
- Strengthening of post-donation information, particularly during the 28 days following the donation when the donor is required to inform the blood center generally of any signs of influenza and particularly of signs of SARS-Cov-2 infection.
Stock level sufficiency
- Following the stock in LBP to inform the NCBTH in case of shortage.
- Give special attention to critical blood products such as platelets, which have a limited life (5 days only). ? Quarantine of the plasma produced since February 2020.
- The use of the plasma intended for fractionation as therapeutic plasma.
- Continue to recruit blood donors at the RCBT all day long while respecting the restriction measures recommended by the local authorities.
Patient hemovigilance
- Reinforcement of the hemovigilance system at the RCBT level and feedback after blood donation.
- Rigorous traceability of LBPs transfused during this critical period and recording or reporting of any transfusion related adverse reaction.
- Donors should be made aware of the importance of self-reporting in case of discomfort and immediately report any symptoms associated with COVID-19 to the ORCBT within 28 days of donation.
Donor and staff safety
- Regular decontamination and continuous ventilation of premises and offices, wearing of masks, disposable gowns and gloves, regular hand disinfection, and distancing.
- Appointment scheduling at the RCBT to minimize the risk of donor assembly and donor crowding at the RCBT.
- The organization of laboratory activities in such a way as to ensure continuity of work under these critical conditions even in the event of contamination of laboratory personnel.
Blood demands management
- Encourage hospital departments to implement a protocol for disinfecting the package (blood requisitions + samples from suspected or COVID-19 infected patients for pre-transfusion immunohematological testing) before sending it to the RCBT.
- The implementation of a clearly identified circuit for the reception of the package, disinfection, sorting, and classification of the LBP demands.
- Awareness of hospital services in order to optimize the prescription of LBP demands by avoiding excessive demands and focusing on urgent demands.
Number of donors
After the declaration of a state of public health emergency on March 20, 2020, only 7 blood collections were carried out in 2020 in mobile donation sites until May, instead of 30 in 2019 (23 fewer collections). All blood collections outside of Oujda have been cancelled, unlike 12 collections made in 2019 outside of Oujda. However, at fixed sites, there were more donors than at mobile site (Table 1). Thus, despite the 25% drop in the number of blood collections observed since April, the ORCBT was able to recruit 7510 donors at both fixed and mobile sites, compared with 8445 donors in 2019 during the same period. The number of donors has generally shown a manageable decrease in 2020 with a drop rate of 11% (Table 1).
LBP demands
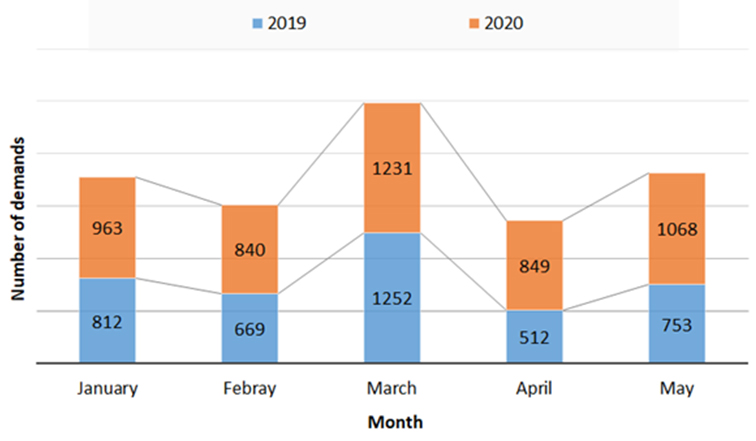
We noted a 23% increase in the number of LBP demands received by the ORCBT between 2019 and 2020 with 3998 demands in 2019 and 4951 demands in 2020 (Figure 1). An increase is most noticeable in the month of March of both years with a minor difference between the two years. A drop in LBPs demand was observed in April 2020, largely due to a reduction in hospital admissions. Average daily LBP demands are estimated at 27 demands per day in 2019 compared to 33 demands in 2020.
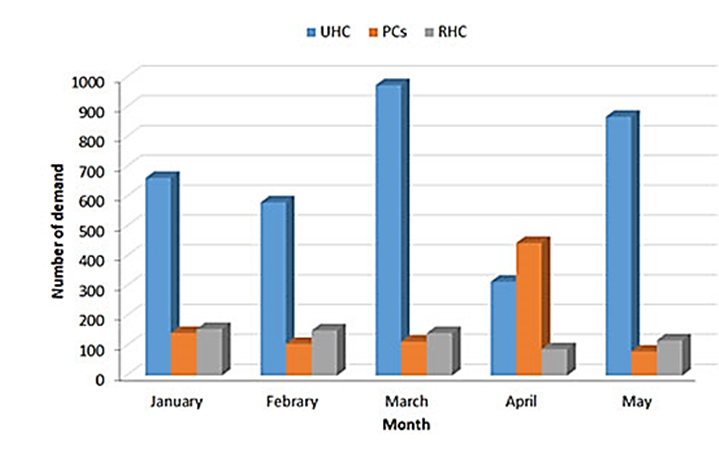
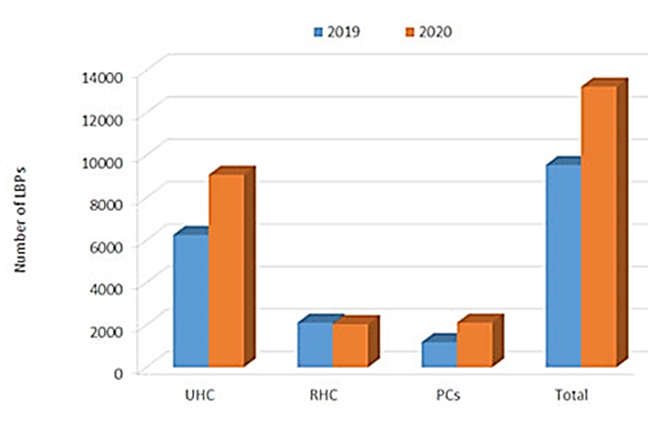
The University Hospital Center (UHC) took the lion’s share of LBP demands by registering 9081 prescribed LBP demands in 2020 compared to 6244 demands in 2019 (Figure 2).
LBPs distribution
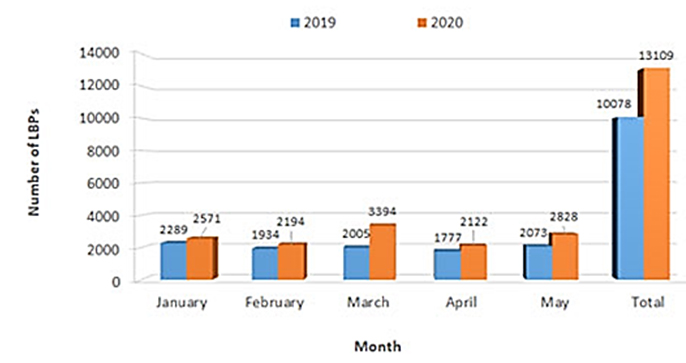
The ORCBT is a regional structure that responds to the LBP needs of the health care facilities within its territory, including the University Hospital Center (UHC), the 7 Regional Hospital Centers (RHC), and the private clinics (PCs) in the region. During the first five months of the year, the ORCBT delivered 13,109 LBPs in 2020, compared with 10,078 LBPs delivered in 2019 during the same period, with an increase of 30% (+3031 LBPs) distributed, as shown in Figure 3.
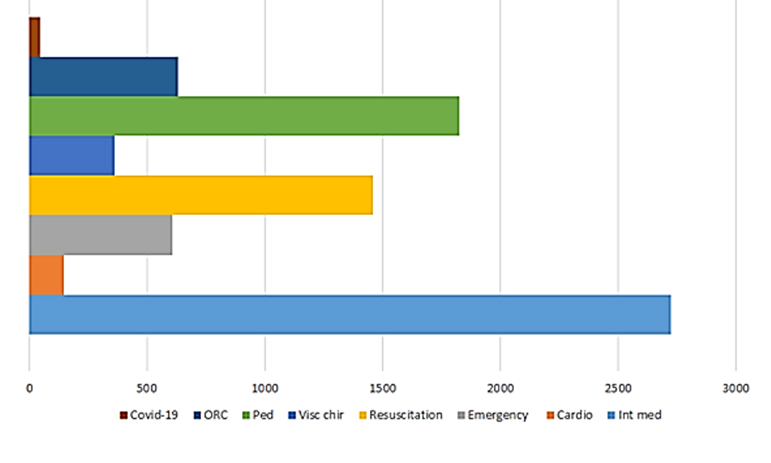
The Department of General Internal Medicine topped the list of services that consumed the most LBPs during the pandemic period (2726), followed by pediatrics (1825), resuscitation (1459), regional oncology (632), and emergency (606) (Figure 4).
It should be noted that an estimated 47 LBPs were delivered to COVID-19 patients for possible transfusion (Figure 4). This number may be underestimated as some of these patients may have been transfused without being declared COVID-19 at the Blood Center.
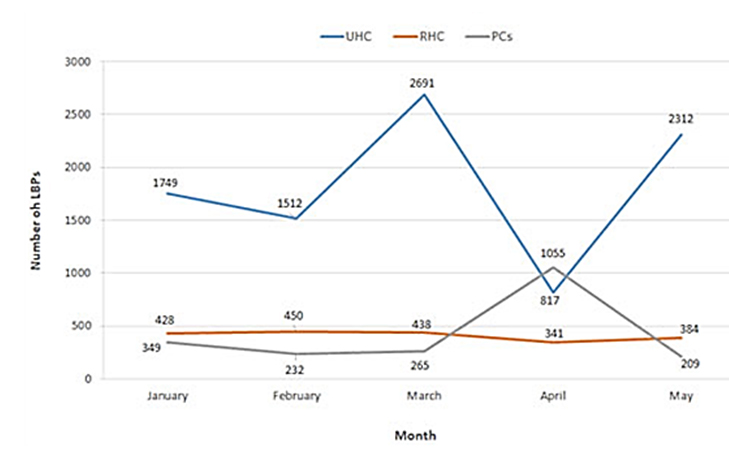
Like all public health facilities in the country, the Al Farabi Regional Hospital Center (RHC), during the pandemic, restructured its services by grouping medical and/or surgical departments separately from the COVID-19 circuit in order to ensure continuity of services despite the hospitalization of COVID-19 patients. At the same time, the number of LBPs delivered to the various RHC services in 2020 decreased compared to 2019, with 2102 in 2020 and 1873 LBPs in 2019 (Figure 5). Hospital services received 94% of the LBPs, mainly in the internal medicine department (509), the emergency department (374), gynecology (212), resuscitation (104), and the nephrology department (67). On the other hand, and exceptionally, Private Clinics (PCs) have registered a considerable fourfold increase in the number of LBPs delivered in April 2020 (Figure 6). For the public healthcare facilities of the cities of the eastern region, the RHC of Nador is at the top of the list of consumers in the LBPs (209), followed by Berkane (189), Jerrada (80), and Guercif (41).
Satisfaction rate of LBP demands
The 2020 application satisfaction rate calculated by type of LBP shows an improvement over the previous year especially for Standard Platelet Concentrates (SPC) as shown in Table 2. 90.49% of demands received from the University Hospital are satisfied for all types of LBP, compared to 81.26% in 2019. In general, there was a marked improvement between 2019 and 2020. The same is true for the Al Farabi RHC and PCs in the region, whose satisfaction rate in 2020 is 85% and 92.46%, respectively, compared to 80.50% and 87.94% in 2019. It should be noted that limiting the number of hospitalizations at the health facility has helped the center to satisfy the demands in LBP.
Stock status
Based on this situation marked by an increase in the number of demands and LBP delivered and a decrease in the number of donors during the month of March and April of the year 2020, we have evaluated the status of the supply at the ORCBT during the pandemic period compared to the previous year.
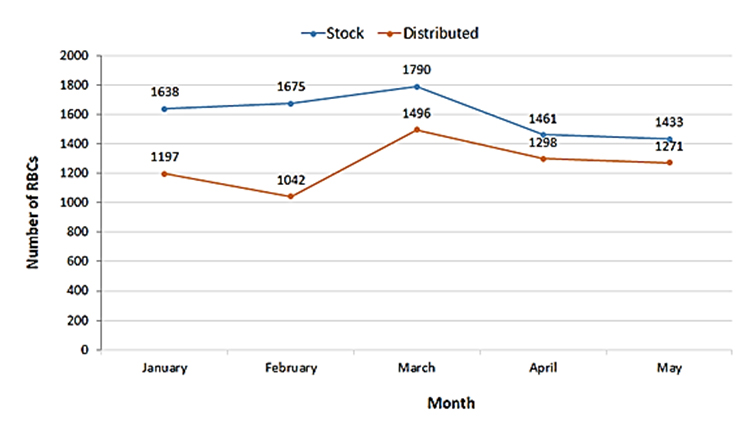
The results of this evaluation showed that the number of RBCs in stock at the beginning of April reached 1298 RBCs exceeding twice the minimum stock (minimum stock 500 units), allowing the ORCBT to satisfy the majority of the RBCs demands addressed to it. However, at the end of May this stock was depleted, reaching 275 RBCs only at the end of May 2020 (Figure 7).
Discussion
With the declaration by WHO of Coronavirus 2019 (SARS-CoV-2) as a pandemic [1], global health care faced unprecedented challenges with rapid and widespread person-to-person transmission of SARS-CoV-2 and high morbidity and mortality worldwide. This has led to an acute worldwide shortage of not only hand disinfectants, personal protective equipment, ventilators, and hospital beds, but also of supplies of LBPs [21], as these are considered essential medicines [9], and are one of the six elements of a health care system. In fact, to satisfy a country’s blood needs, it is recommended that there should be >1% donors in ratio to the general population [22]. In Morocco, before the declaration of the state of health emergency, the number of donors was estimated at 0.96% of the Moroccan population. This percentage remains low compared to the WHO recommendations (1.2% for developing countries), indicating that Morocco is below the norm.
Two months after the start of the pandemic in the city of Wuhan, the number of donors increased considerably, especially in February and March compared to the previous year. This is due to the proactive measures implemented as early as February by the ORCBT to anticipate the crisis. Indeed, in the absence of COVID-19 cases in the kingdom, the strengthening of mobile collections, coordination with hospital services to optimize LBP demands, and the use of social networks were able to increase the number of donors.
As of March 20, 2020, the date of the declaration of the state of health emergency, we noted a 50% drop in the number of voluntary donors in mobile sites due to the individual confinement measures imposed by the Moroccan authorities. This decrease led to the cancellation of 10 collections on average in mobile site, correlated to a loss of 1026 donors on average, which corresponds to the loss of 3080 LBPs per month. This impact of the pandemic on blood collection has been seen in other countries [21],[23],[24]. In the United States, as a result of assembly restrictions, nearly 4000 American Red Cross blood collections were cancelled across the country [21]. These cancellations resulted in some 130,000 fewer blood donations in just a few weeks. Previously, results have been observed during the H1N1 pandemic, which had a significant impact on the blood supply due to donors’ fear of being exposed to the virus in a hospital or independent blood donation facility [16]. In ordinary situations, the number of donors at fixed sites was always almost inferior to the number of donors at mobile sites, a finding noted in other countries [21] where the majority of blood collected comes from non-permanent collection sites. However, after the declaration of the health emergency, the ORCBT registered more donors at fixed sites than at mobile sites. Indeed, the strategy implemented by the ORCBT, including coordination with local associations and social networks, especially Facebook groups to encourage their subscribers to donate blood by bringing them back to the center by group and also by scheduling appointments by phone or via Google doc while respecting the hygiene measures in vigor, has made it possible to recruit more than 1250 fixed-site donors, compared to 864 donors in 2019 during April and May. In addition, in the month of Ramadan, during the daytime, blood collections at the fixed site are suspended and usually organized at night at the mosques after the “Taraouih” prayers. This year, this month has coincided with the pandemic period when night movements are banned. As a result, the ORCBT has found it more difficult to recruit blood donors at both fixed and mobile sites. Through the strategy described above, and in particular the authorization granted by the authorities and the collaboration with civil society and the provision of a means of transport for donors, the center has managed to organize 14 night collections, including 7 mobile collections organized with local authorities and health organizations in the eastern region, in addition to 7 night collections carried out at the ORCBT. The number of LBPs delivered has generally increased by 30%, especially in the month of March in anticipation of medical programs before school holidays according to the Moroccan program. The surprising thing is that private clinics recorded a number of demands for LBP multiplied by 4 during the month of April in an unusual way, this action is justified by the migration of patients to these establishments for fear of contamination in the public structures receiving COVID-19 patients. Given that the epidemiological situation up to the end of May is different in comparison with other sites [25], particularly in the eastern region (7780 cases and 204 deaths, including 187 cases and 8 deaths in the eastern region on 31 May), our results differed from those found in the United States of America and Singapore [15],[26], where the overall use of LBPs decreased significantly for hospitalized patients. In fact, during a pandemic period when the number of COVID-19 positive cases is high and consequently there is an increased fear of contamination, the number of routine surgeries has decreased due to the postponement of elective surgeries and the transfusion of LBPs to hospitalized patients has practically decreased. The LBP distributions by department show that the Internal Medicine and Pediatrics Department was the first to procure LBPs (Figure 4). This is mainly due to the frequent hematological pathologies among patients in these services requiring transfusion as a therapeutic procedure (Leukemia, anemia, etc.) [27]. An exception was noted regarding LBP demands for patients in intensive care with COVID-19. This finding was reported in a study [28] in which blood demands were provided for COVID-19 patients admitted to the intensive care unit in 96.1% with lymphocytopenia, 57.7% with thrombocytopenia and 61.1% with leukopenia. In fact, the study by Wang et al. [29] showed that patients who were admitted to intensive care units had a higher number of comorbidities (72.2%)than those who were not admitted to this service (37.3%). This suggests that comorbidities, such as hematological pathologies, may be risk factors.
Regarding all of the above, the proactive measures based on the reinforcement of collections since February as well as the strategy implemented during the pandemic, have permitted the RCBT to have a sufficient stock of LBP to satisfy the needs of health care facilities in the region with a satisfaction rate of more than 90%. Similar measures were used in China during the pandemic. However, in the early stages of the epidemic where it is unclear or even unknown how COVID-19 is transmitted to populations, nearly 80% of people mobilized via text message feared SARS-CoV-2 contamination during the donation process and were less willing to donate blood. The success rate of recruitment via SMS fell by 60%. The number of all blood donors decreased rapidly by 60–70% [30].
Conclusion
Recent studies have shown that the COVID-19 epidemic itself did not lead to an extreme increase in blood demand, but to a shortage of blood caused by a drop in the number of donors. Donors feared contamination by the virus during movement to collection centers and at the moment of blood donation. This study shows that, to some extent, the RCBT’s proactive strategy overcame this problem. This is due to the proactive measures that were implemented by the OHRTC and the accompanying directives from the NCBTH. This experience has been documented to serve as a possible response model to develop emergency plans and to generate to other services. In summary, an effective strategy has to be implemented in blood banks for possible crises where the development of permanent communication tools, the improvement of these techniques including the discourse adopted to convince donors, the improvement of LBP management and the optimization of its use are the essential points to be taken seriously.
REFERENCES
1.
Novel Coronavirus – China. 2020. [Available at: https://www.who.int/csr/don/12-january-2020-novel-coronavirus-china/en/]

2.
World Health Organization. Coronavirus disease (COVID-19) Situation Report – 157. Health World Organization. 2020. [Available at: https://apps.who.int/iris/handle/10665/332857]

3.
4.
COVID-19 WHO African ragion External Situation Report 17. 2020. [Available at: https://apps.who.int/iris/bitstream/handle/10665/332705/SITREP_COVID-19_WHOAFRO_20200624-eng.pdf]

5.
WHO Director-General’s opening remarks at the media briefing on COVID-19 -11 March 2020. 2020. [Available at: https://www.who.int/director-general/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020]

6.
Africa COVID-19 cases top 100 000. 2020. [Available at: https://www.afro.who.int/news/africa-covid-19-cases-top-100-000]

7.
WHO ramps up preparedness for novel coronavirus in the African region. 2020. [Available at: https://www.afro.who.int/news/who-ramps-preparedness-novel-coronavirus-african-region]

8.
Chan JFW, Yuan S, Kok KH, et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: A study of a family cluster. Lancet 2020;395(10223):514–23. [CrossRef]
[Pubmed]

9.
Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020;395(10223):497–506. [CrossRef]
[Pubmed]

10.
Drosten C, Günther S, Preiser W, et al. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N Engl J Med 2003;348(20):1967–76. [CrossRef]
[Pubmed]

11.
Cho HJ, Koo JW, Roh SK, et al. COVID-19 transmission and blood transfusion: A case report. J Infect Public Health 2020;13(11):1678–9. [CrossRef]
[Pubmed]

12.
Chang L, Yan Y, Wang L. Coronavirus disease 2019: Coronaviruses and blood safety. Transfus Med Rev 2020;34(2):75–80. [CrossRef]
[Pubmed]

13.
AABB. Coordinating the nation’s blood supply during disasters and biological events. www.aabb.org: AABB; 2008.

14.
Products NACOBB, Services CB. The National Plan for Management of Shortage of Labile Blood Components. National Advisory Committee on Blood & Blood Products Canadian Blood Services; 2020. Contract No.: 2. https://www.nacblood.ca

15.
Pagano MB, Hess JR, Tsang HC, et al. Prepare to adapt: Blood supply and transfusion support during the first 2 weeks of the 2019 novel coronavirus (COVID-19) pandemic affecting Washington State. Transfusion 2020;60(5):908–11. [CrossRef]
[Pubmed]

16.
WHO. Maintaining a safe and adequate blood supply during the pandemic outbreak of coronavirus disease (COVID-19). WHO; 2020. Contract No.: 1. [Available at: https://www.who.int/publications]

17.
WHO. Guidance on maintaining a safe and adequate blood supply during the coronavirus disease 2019 (COVID-19) pandemic and on the collection of COVID-19 convalescent plasma. WHO; 2020. [Available at: https://www.who.int/publications]

18.
Santé Mdl. Stratégie nationale du dépistage du COVID-19. Ministère de la Santé; 2020. Report No.: 037DELM/00. [Available at: https://fesnews.net/wp-content/uploads/2020/05/Stratégie-nationale-du-COVID-19.pdf]

19.
20.
Transfusion sanguine et Pandémie Covid-19: Recommandations et mesures prises par la Centre National de Transfusion Sanguine et d’Hématologie. 2020. [Available at: https://covidaba.com/en/transfusion-sanguine-et-pandemie-covid-19-recommandations-et-mesures-prises-par-le-centre-national-de-transfusion-sanguine-et-dhematologie/]

21.
22.
Vers 100% de dons de sang volontaires: Cadre mondial d’action. 2010. [Available at: https://www.who.int/publications/list/9789241599696/fr/]

23.
24.
25.
WHO. Situation Report – 132. WHO; 2020. [Available at: Available at: https://www.who.int

26.
Fan BE, Ong KH, Chan SSW, et al. Blood and blood product use during COVID-19 infection. Am J Hematol 2020;95(7):E158–60. [CrossRef]
[Pubmed]

27.
Renaudier P. Transfusion in haematology. [Article in French]. Transfus Clin Biol 2008;15(5):228–35. [CrossRef]
[Pubmed]

28.
Terpos E, Ntanasis-Stathopoulos I, Elalamy I, et al. Hematological findings and complications of COVID-19. Am J Hematol 2020;95(7):834–47. [CrossRef]
[Pubmed]

29.
Wang D, Hu B, Hu C, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China 2020;323(11):1061–9. [CrossRef]
[Pubmed]

30.
Wang Y, Han W, Pan L, et al. Impact of COVID-19 on blood centres in Zhejiang province China. Vox Sang 2020;115(6):502–6. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Acknowledgments
We salute the efforts of all health care workers and especially the personnel of ORCBT during this pandemic. We warmly thank all the donors and BDAER who helped us to overcome this crisis.
Author ContributionsSaliha Mharchi - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Mounir Cherfi - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ahmed Karim - Acquisition of data, Analysis of data, Drafting the work, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Abdelhamid Bouazza - Acquisition of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Zaina Sidqi - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Mohammed Benajiba - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2021 Saliha Mharchi et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.